Prototype tooling for quick components testing
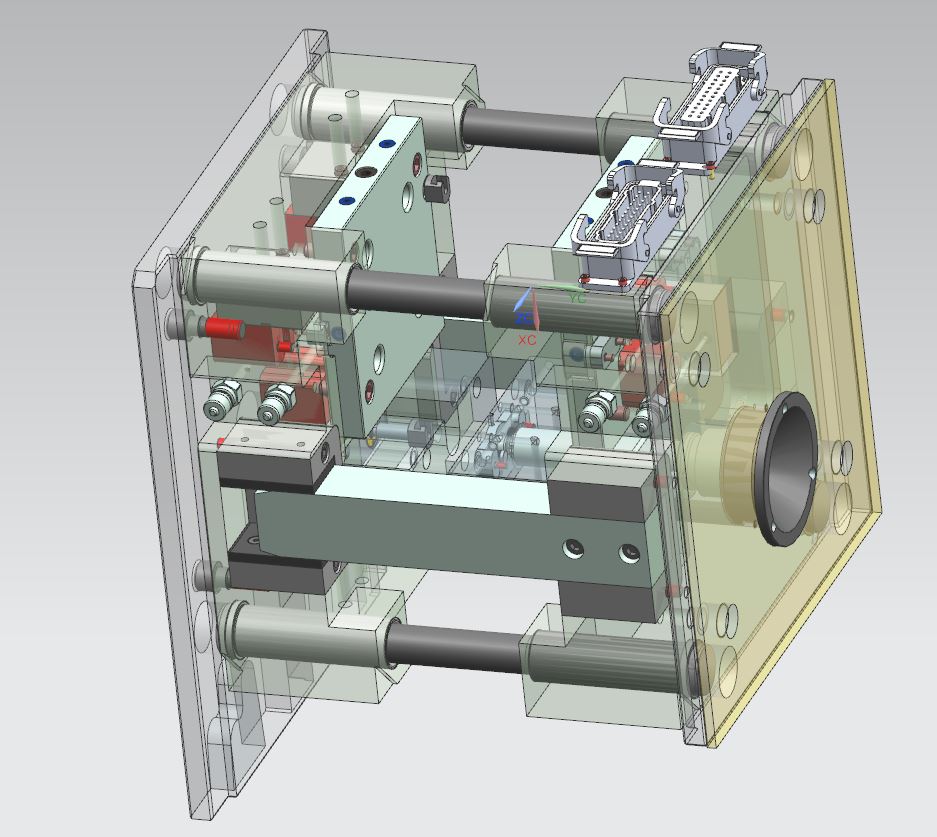
In IVD and medical device component development, the need of validation of the designs quickly is a key to make strong steps forward.
The parts to be tested will need to meet the requirement of being manufactured in conditions as close as possible as the final product manufacturing, due to the precision and repeatability that are necessary in these kind of IVD or lab on chip products.
For that purpose, it is important to work in two areas to achieve this goal:
Infrastructure: Adequate toolbase with the maximum of a final tool needed for these kind of parts integrated on it. Quick inserts adaption and fixing system will help to reduce mounting / dismounting time, always keeping in mind an accurate positioning of the inserts for a perfect repeatability of the process.
Standards definition: It´s important to define several parameters of the component as standard to be able to reuse mould areas and minimize changes, this will help a lot to reduce manufacturing time.
Defining a standard tool, with defined inlets, outlets, external size, ejectors position, etc…will be important to get quick iterations, of components sections, to validate the product before the final tool with the final optimized design.
If you have enquiry about this issue dont hesitate to contact us sending an email to medical@erreka.com or by fullfilling the form located at the contact area of the website.
News
We tell you the latest news about ERREKA.

Polymer Science: Driving Innovation in Health and Industry
Polymer science enables the production of devices for a wide range of applications, including biomedical diagnostic (such as point-of-care diagnostics, […]
IVD Raw materials
With the enhancement of medical science and technology, there’s a growing need for real-time diagnostic devices. Many compact systems have […]

From glass chips to polymer chips: revolutionizing microfluidic technology
For many years, glass chips have been integral to biomedical applications, notably in microfluidic technology. However, recent advancements have illuminated […]